 Specific gravities for common fluids like acetone, alcohol, turpentine, oil and more. Variations in jet fuel density as function of temperatur, together with volume correction factors.ĭensities of common liquids like acetone, beer, oil, water and more. Hydrocarbons, Linear Alcohols and Acids - Densitiesĭensity of hydrocarbons like alcohols and acids as function of carbon number at 20☌ / 68°. Molweight, melting and boiling point, density, flash point and autoignition temperature, as well as number of carbon and hydrogen atoms in each molecule for 200 different hydrocarbons. Variations in fuel oils density as function of temperatur, together with volume correction factors. Specific Weight and Specific GravityĪn introduction to density, specific weight and specific gravity. Density of inorganic substances in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensity vs. Density of some sugars, alcohols and other organic substances in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensity of Aqueous Solutions of some Inorganic SubstancesĬhanges in density of aqueous solutions with changes in concentration at 20☌. Density of acetic acid, citric acid, formic acid, D-lactic acid, oxalic acid and trichloroacetic acid in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensity of Aqueous Solutions of Organic Substances as Sugars and AlcoholsĬhanges in density of aqueous solutions with changes in concentration at 20☌. Density of inorganic sodium salts in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensities of Aqueous Solutions of Organic AcidsĬhanges in density of aqueous solutions with changes in concentration at 20☌. Density of potassium salts in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensities of Aqueous Solutions of Inorganic Sodium SaltsĬhanges in density of aqueous solutions with changes in concentration at 20☌. Density of inorganic chlorides in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensities of Aqueous Solutions of Inorganic Potassium SaltsĬhanges in density of aqueous solutions with changes in concentration at 20☌. Hydrodynamic losses through pumps depends on fluid viscosities.ĭensities of Aqueous Solutions of Inorganic ChloridesĬhanges in density of aqueous solutions with changes in concentration at 20☌. Online calculator, figures and table showing dynamic and kinematic viscosity of benzene, C 6H 6, at varying temperature and pressure - Imperial and SI Units. Specific Gravity.īenzene - Dynamic and Kinematic Viscosity vs. Molweight, melting and boiling point, density, pKa-values, as well as number of carbon and hydrogen atoms in molecules are given for 150 different alcohols and acids. Maximum distance between supports for ABS, PE, PVC, Fiberglass and Steel Pipes.Īlcohols and Carboxylic Acids - Physical Data Material properties of gases, fluids and solids - densities, specific heats, viscosities and more.ĪBS, PE, PVC, Fiberglass and Steel Pipes - Supports Pipe lines - fluid flow and pressure loss - water, sewer, steel pipes, pvc pipes, copper tubes and more. Just above and below 4☌ they are almost the same.Densities of solids, liquids and gases. 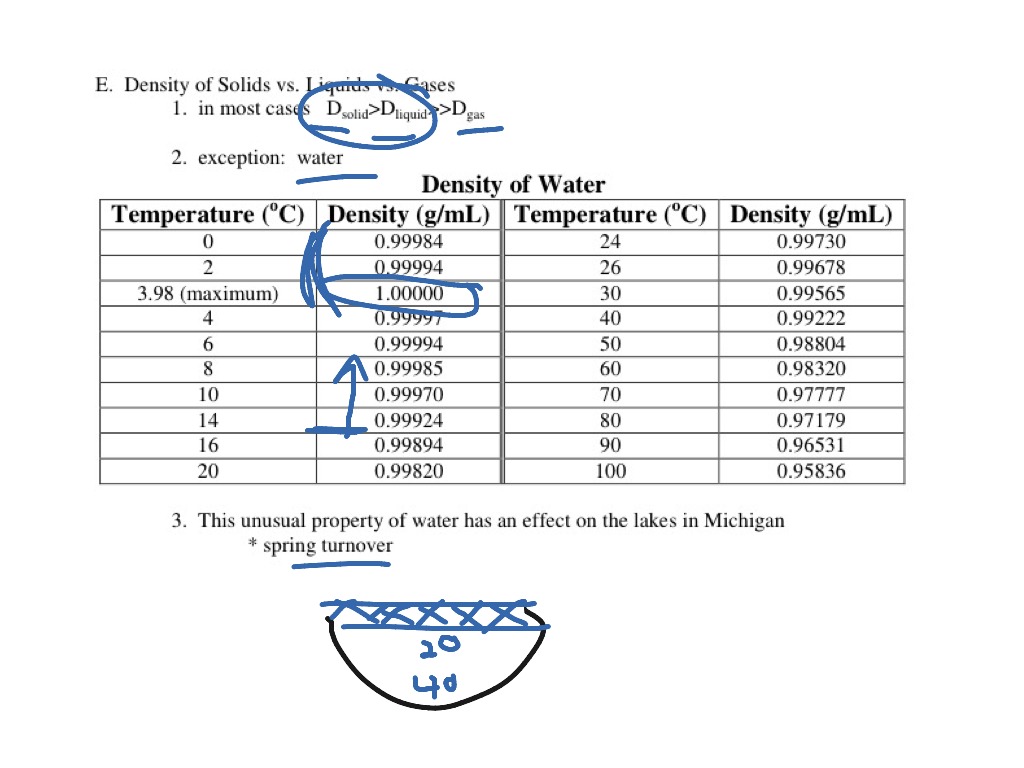
Water samples just above and below 0☌ are very different. At that T, the tendency toward settling into a special low-energy structure runs away in a cooperative manner, and ice crystals form. 
4☌ just happens to be the T at which the expansion, due to settling into the special low-energy states, and the contraction, due to the general trend to spend more time in contact at low T, cancel.Ġ☌ is another matter. Nothing really special happens there in the structure of the water. At higher T, the molecules sample even higher energy states, and a majority of them are loosely packed, so the liquid expands- the more typical behavior. When things cool, they settle into lower energy arrangements. For most molecules, that typically involves squeezing together a bit, but water happens to have a low-energy arrangement that’s rather open. 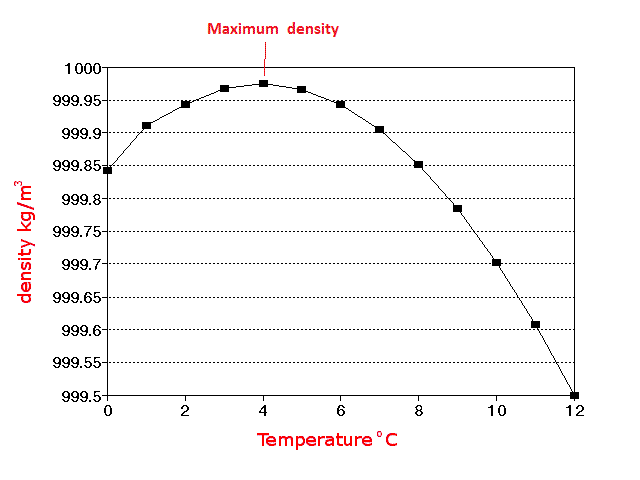
The expansion of water at lower T results from the water molecules arranging themselves to minimize the energy of their interactions. I assume you are referring to liquid water, not ice, since 4☌ is about the temperature (T) at which liquid water has a minimum volume, at atmospheric pressure. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
